Biomolecules and their structural characteristics.
The water molecule has a significant tendency to ionize into a hydroxide ion (OH-) and a hydrogen ion (H+). Actually two water molecule
participate in this ionisation.
participate in this ionisation.
The hydronium ion is in effect a hydrogen ion bound to a water molecule. The ionisation of water is important to all living creatures. pH is a measurement of hydrogen concentration. How acidic or basic a solution is depends on the relative concentrations of H+ and OH- ions on it. A water molecule is made from one oxygen ion and two hydrogen atoms, bounded
together by covalent bonds.
The oxygen atoms has the ability to attract the negative electrons away from the two hydrogen atoms. This type of covalent bonding is called a
polar bond. Water is often referred as the universal solvent because it can dissolve so many chemicals. The polarity of the water molecule allows water to
dissolve other chemicals that have charges to them. Most common example is salt or sodium chloride. Sodium chloride contains positive sodium cations and
negative chlorides anions.
Carbohydrates
All living things consist of simple substances called elements. Living organisms are made up of a limited number of atoms that combine to from molecules.
Carbon is and element that is found in all organic molecules. The term organic means living. The carbon atoms can join together With carbon-carbon bonds forming chains or rings that are the skeletons of organic molecules and thus life.
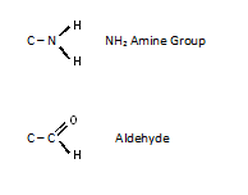
Groups of other elements can be attached at various positions of the carbon skeleton. Each group has its own properties. The COOH group is responsible for the acidic nature of fatty acids and amino acids. Carbon also has the ability to form multiple carbon bonds with other carbon atoms, oxygen and nitrogen.
C – X (halogen) = Halide
C – OH = alchohol
There are three important types of macromolecules in biology, namely:
- Polysaccharides (consist of monomers)
- Proteins
- Nucleic acids
Carbohydrates
Carbohydrates have many functions in organisms. Carbohydrates are used as; energy sources (glucose is used in respiration), energy storage (glycogen is stores in the liver and skeletal muscles in mammals), building blocks for the biosynthesis of cellular components, structural molecules (cellulose), information molecules (ribose is used in RNA).
The functional parts played by carbohydrates in plants are played by proteins in animals. The general formula for a carbohydrate is Cn(H2O)n1.
The most common energy source in the body is a sugar called glucose. Glucose releases energy from its bonds when respiration occurs.
Glucose + oxygen ----> Carbon dioxide + water + energy
The energy stored in a chemical called adenosine triphosphate (ATP), which can be used by cells as an energy supply.
Carbohydrates are all aldehydes or ketones .Carbohydrates also contain several hydroxyl groups. Carbohydrates can be classified as: Monosaccharides (Containing one sugar unit; glucose and fructose), disaccharides (containing two sugar units eg; sucrose, maltose and lactose), triaccharides (containing three sugar units), Oligosaccharides (containing between 4 and 10 sugar units), polysaccharides (Containing more than ten sugar units).

