Bond Energy
Bond making ---> one would have energy being released with bond making therefore it is exothermic.
Bond breaking ---> one needs energy to break a bond therefore it is endothermic.
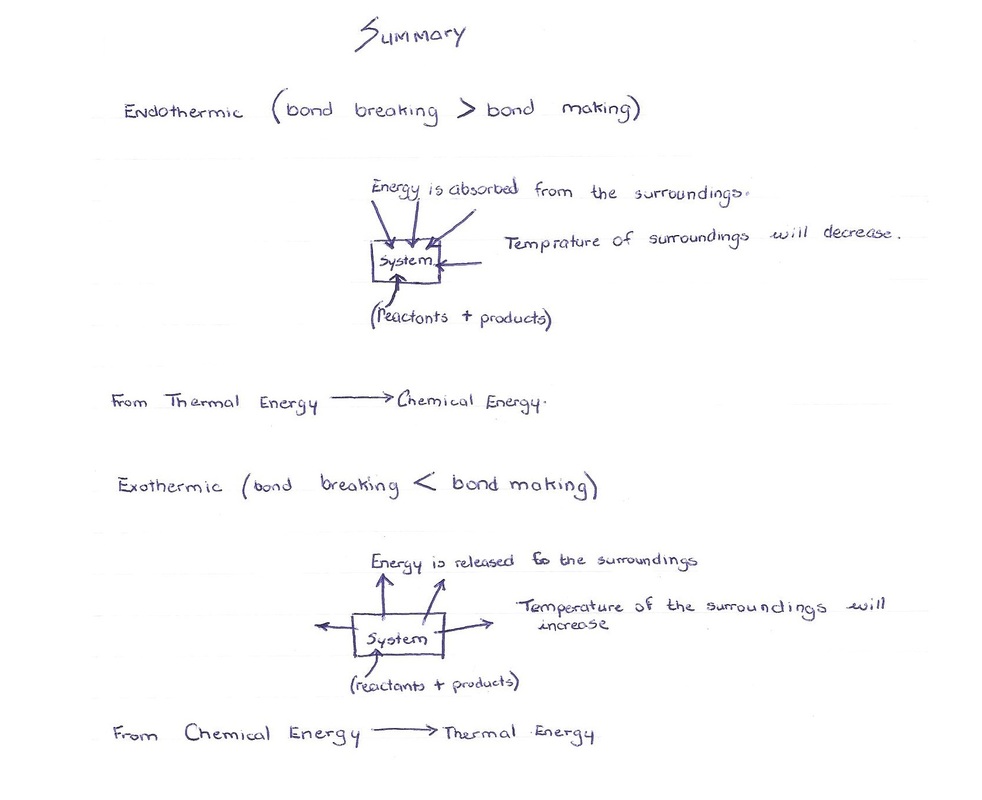
A reaction may be overall endothermic or exothermic. Endothermic also means that overall more bonds are being broken than made. Exothermic also means that overall more bonds are being made than broken.
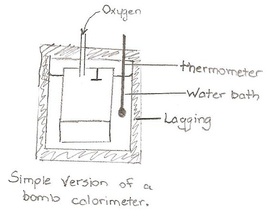
The study that involves exothermic or endothermic energy is called thermochemistry. Thermochemistry is the study of heat of the reaction also known as enthalpy. Exothermic has a negative enthalpy. Negative enthalpy is when energy is released from the system (reactants + product) to the surrounding. Endothermic has a positive enthalpy. It is positive because the system absorbs energy from the surroundings. To measure the change (i.e. endothermic or exothermic) one may use temperature.
Systems and Surroundings
There are three types of systems:
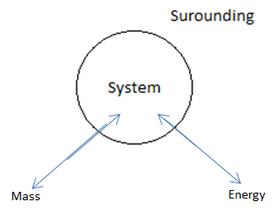
1 – Open system: Lets the transfer of both mass and energy.
2 – Closed system: Lets the transfer of energy but not mass.
3 – Isolated: Lets no mass or energy transfer.
2 – Closed system: Lets the transfer of energy but not mass.
3 – Isolated: Lets no mass or energy transfer.
Enthalpy = ∆H
Standard Enthalpy = ∆Hᶲ
Unit = kJ mol-1
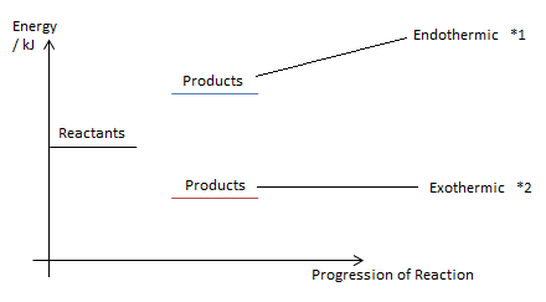
One needs to use a closed system to calculate enthalpy. Energy profiles (it involves only the system).
*1 Products will have less energy than reactants. It releases energy to the surroundings.
*2 Products have higher energy than reactants. It gains energy from the surroundings.
Standard Enthalpy = ∆Hᶲ
Unit = kJ mol-1
One needs to use a closed system to calculate enthalpy. Energy profiles (it involves only the system).
*1 Products will have less energy than reactants. It releases energy to the surroundings.
*2 Products have higher energy than reactants. It gains energy from the surroundings.
Standard enthalpy change of combustion: is the energy released when 1 mole of substance reacts completely with oxygen at atmospheric pressure.
∆Hcᶲ C(s) + O2 (g) ---> CO2 (g)
Standard enthalpy of neutralization is the energy given off when 1 mole of water is produced when an acid and a base react at atmospheric pressure and temperature.
∆Hcᶲ C(s) + O2 (g) ---> CO2 (g)
Standard enthalpy of neutralization is the energy given off when 1 mole of water is produced when an acid and a base react at atmospheric pressure and temperature.