Chemistry for Biology Technicians
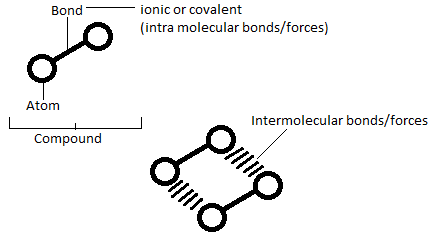
Intramolecular forces / bonds
- Ionic Bonding
Ionic bonding is the bonding that results when a ‘metal’ atom looses electrons (e-) to a ‘non-metal’ atom that accepts these electrons. Therefore the bond would be made of a electrostatic forces between a positive ion/s and negative ion/s.
Note: Positive ion = cation
Negative ion = anion
- Convalent Bonding
A covalent bond is the bond created when 2 or more ‘non-metal’ atoms share their electrons, so to complete their outer shell. Covalent bonds are stronger to break then ionic bonds.
Intermolecular forces / bonds
Intermolecular forces are the electrostatic forces between different or same compounds. There are three levels of forces depending on the polarity (electronegativity) of the atoms being envolved. (research Pauling’s scale)
The three forces are:
- Van der Waal’s forces
- Dipole-dipole forces
- Hydrogen bonds
Van der Waal’s forces: these are the weakest forces one can find between compounds since they are created when momentarily (temporarily) dipoles are created within a molecule.
Dipole-dipole forces: These are stronger then Van der Waal’s. Dipole-dipole happens between compounds that have partially charged (Small delta sign) atoms.
- Ionic Bonding
Ionic bonding is the bonding that results when a ‘metal’ atom looses electrons (e-) to a ‘non-metal’ atom that accepts these electrons. Therefore the bond would be made of a electrostatic forces between a positive ion/s and negative ion/s.
Note: Positive ion = cation
Negative ion = anion
- Convalent Bonding
A covalent bond is the bond created when 2 or more ‘non-metal’ atoms share their electrons, so to complete their outer shell. Covalent bonds are stronger to break then ionic bonds.
Intermolecular forces / bonds
Intermolecular forces are the electrostatic forces between different or same compounds. There are three levels of forces depending on the polarity (electronegativity) of the atoms being envolved. (research Pauling’s scale)
The three forces are:
- Van der Waal’s forces
- Dipole-dipole forces
- Hydrogen bonds
Van der Waal’s forces: these are the weakest forces one can find between compounds since they are created when momentarily (temporarily) dipoles are created within a molecule.
Dipole-dipole forces: These are stronger then Van der Waal’s. Dipole-dipole happens between compounds that have partially charged (Small delta sign) atoms.
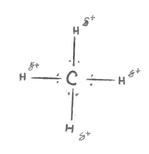
Hydrogen bonding is the strongest intermolecular force. This force is created between compounds that have hydrogen covalently bound to oxygen, nitrogen and fluorine.
H –F (hydrogen floridide)
H – O (water, hydrogen peroxide, alchohols, carboxylic acids)
H – N (Ammonia, urea, Amines, Amides)
Note: Intermolecular forces can have large influences on the melting point and boiling point of a compound.
Hydrophobic and Hydrophilic compounds
A hydrophobic compound will try to minimize the contact with water. A hydrophilic compounds will maximize it’s contact with water.
__________________________________________________________________________________________________________________________________
Substances
Salt: is an ionic substance, it easily forms ions when dissolved in water.
Solution: is when a solute dissolves in a solvent
Solids: have particles closer to each other with the least kinetic energy.
Liquids: are substances that have more kinetic energy than solids but less than gases.
Gases: have the most kinetic energy, as a matter of fact in fact they can freely move around.
Emulsion: is when two liquids can be mixed together but do not dissolve in each other.#
Bonding vs. Physical Properties
Bond making ---> energy given off by the system (substance)
Bond breaking ---> energy being absorbed by the system (substance)
Bonding being intramolecular or intermolecular will have an effect on the physical properties (characteristics). The intramolecular (ionic and covalent) will determine the solubility. An ionic substance is more soluble than covalent. Since ionic bonds are electrostatic forces, hence weaker than covalent bonds, they are easier to break. The intermolecular bonds will determine the ‘change of state’ i.e. the melting point and boiling point. Those compounds that have Van Der Waal’s have lower melting points and boiling points than hydrogen bonding.
H –F (hydrogen floridide)
H – O (water, hydrogen peroxide, alchohols, carboxylic acids)
H – N (Ammonia, urea, Amines, Amides)
Note: Intermolecular forces can have large influences on the melting point and boiling point of a compound.
Hydrophobic and Hydrophilic compounds
A hydrophobic compound will try to minimize the contact with water. A hydrophilic compounds will maximize it’s contact with water.
__________________________________________________________________________________________________________________________________
Substances
Salt: is an ionic substance, it easily forms ions when dissolved in water.
Solution: is when a solute dissolves in a solvent
Solids: have particles closer to each other with the least kinetic energy.
Liquids: are substances that have more kinetic energy than solids but less than gases.
Gases: have the most kinetic energy, as a matter of fact in fact they can freely move around.
Emulsion: is when two liquids can be mixed together but do not dissolve in each other.#
Bonding vs. Physical Properties
Bond making ---> energy given off by the system (substance)
Bond breaking ---> energy being absorbed by the system (substance)
Bonding being intramolecular or intermolecular will have an effect on the physical properties (characteristics). The intramolecular (ionic and covalent) will determine the solubility. An ionic substance is more soluble than covalent. Since ionic bonds are electrostatic forces, hence weaker than covalent bonds, they are easier to break. The intermolecular bonds will determine the ‘change of state’ i.e. the melting point and boiling point. Those compounds that have Van Der Waal’s have lower melting points and boiling points than hydrogen bonding.
