Rate of Reaction (Chemistry Summary Notes)

Ø The rate of reaction is a measure of how fast or slow a reaction is. Rate is measure if the change that happens in a single unit of time.
Ø The rate of reaction can be altered by:
- Changing the surface area of a solid.
- Changing the concentration of the reactants.
- Changing the temperature of the reaction.
- The presence of light
- Adding a catalyst
- Changing the pressure of gases in gaseous reactions.
Ø Collision theory: the particles have to collide in order to react but they have to have enough energy. The rate of reaction depends on how often and how hard the particles hit each other.
1. Changing the surface area of a solid
Ø The rate of reaction can be altered by:
- Changing the surface area of a solid.
- Changing the concentration of the reactants.
- Changing the temperature of the reaction.
- The presence of light
- Adding a catalyst
- Changing the pressure of gases in gaseous reactions.
Ø Collision theory: the particles have to collide in order to react but they have to have enough energy. The rate of reaction depends on how often and how hard the particles hit each other.
1. Changing the surface area of a solid
2. Changing the concentration of the reactants
3 Temperature if reaction
- In the reaction between hydrochloric acid and sodium thiosulfate, warming the solution makes the sulfur precipitate faster.
4. Presence of light
5. Use of a catalyst
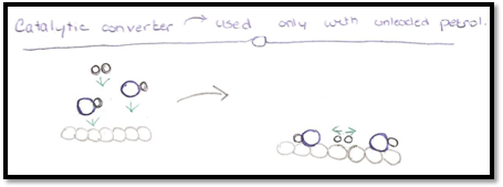
- A catalyst is a substance which although often present in small proportions alters the rate of a chemical reaction, but remains chemically unchanged at the end of the reaction.
- A catalytic converter is used in cars because car exhaust fumes contain pollutant gases such as CO and NO. These are absorbed on to a rhodium catalyst where they react with oxygen to make less harmful products.
2CO (g) + O2 (g) -> 2CO2 (g)
2NO (g) + CO (g) -> N2 (g) + CO2 (g)
3 Temperature if reaction
- In the reaction between hydrochloric acid and sodium thiosulfate, warming the solution makes the sulfur precipitate faster.
4. Presence of light
5. Use of a catalyst
- A catalyst is a substance which although often present in small proportions alters the rate of a chemical reaction, but remains chemically unchanged at the end of the reaction.
- A catalytic converter is used in cars because car exhaust fumes contain pollutant gases such as CO and NO. These are absorbed on to a rhodium catalyst where they react with oxygen to make less harmful products.
2CO (g) + O2 (g) -> 2CO2 (g)
2NO (g) + CO (g) -> N2 (g) + CO2 (g)
