Heat (Physics Summary Notes)
Ø Scales of heat are the Kelvin, Celsius and Fahrenheit scales.
Finding the fixed point of a thermometer: First find the 0oC mark by placing the thermometer into pure melting ice. Then find the 100oC mark by placing the thermometer in a steam chest where pure water is being boiled. Then the scale between them was marked.
Types of thermometers
Ø Liquid in glass thermometer
Ø Bimetallic thermometer
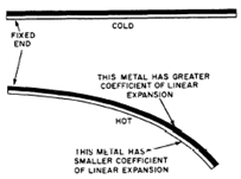
- This type of thermometer is most commonly used in thermostats.
- This thermometer consists of two strips of metals one is copper and one is steel and since copper expands faster than steel so the strip bends.
Specific heat capacity
Ø The specific heat capacity of a substance is defined as the heat energy required to raise the temperature of 1kg by 1oC.
Ø The S.I. unit of specific heat capacity is J/kgoC
Ø The equation for the specific heat capacity is the heat given out equals the mass multiplied by the specific heat capacity multiplied by the temperature change.
- c = specific heat capacity in J/kg*C
- Q = amount of heat energy in J
- m = mass of the substance in kg
- ⍙ø = change in temperature in *C
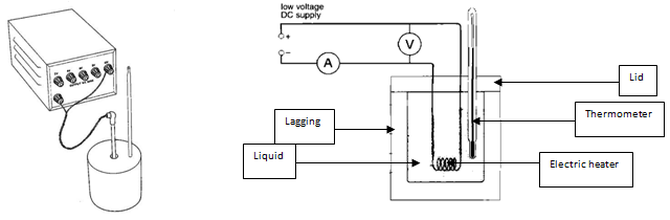
Finding the specific heat capacity of a liquid or solid:
Finding the fixed point of a thermometer: First find the 0oC mark by placing the thermometer into pure melting ice. Then find the 100oC mark by placing the thermometer in a steam chest where pure water is being boiled. Then the scale between them was marked.
Types of thermometers
Ø Liquid in glass thermometer
Ø Bimetallic thermometer
- This type of thermometer is most commonly used in thermostats.
- This thermometer consists of two strips of metals one is copper and one is steel and since copper expands faster than steel so the strip bends.
Specific heat capacity
Ø The specific heat capacity of a substance is defined as the heat energy required to raise the temperature of 1kg by 1oC.
Ø The S.I. unit of specific heat capacity is J/kgoC
Ø The equation for the specific heat capacity is the heat given out equals the mass multiplied by the specific heat capacity multiplied by the temperature change.
- c = specific heat capacity in J/kg*C
- Q = amount of heat energy in J
- m = mass of the substance in kg
- ⍙ø = change in temperature in *C
Finding the specific heat capacity of a liquid or solid:
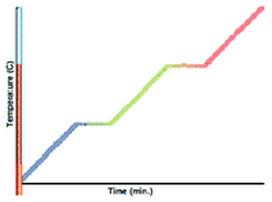
Change of state and latent heat
Ø Matter changes state when heat is applied. This change is from Solid à Liquid à Gas.
Ø When matter is heated it expands and when it is cooled it contracts.
For a Better Explaination go to: http://www.youtube.com/watch?v=1vIF8f1T8a0&feature=related
Evaporating and boiling
Ø Both of the above have the same change of state from a liquid to a gas.
Ø There is a difference between evaporation and the process of boiling. In evaporation the liquid molecules escape without the external heating source but in boiling a heat source is applied so the molecules get the energy to escape.
Effect of impurities on the melting and boiling points
Ø Matter changes state when heat is applied. This change is from Solid à Liquid à Gas.
Ø When matter is heated it expands and when it is cooled it contracts.
For a Better Explaination go to: http://www.youtube.com/watch?v=1vIF8f1T8a0&feature=related
Evaporating and boiling
Ø Both of the above have the same change of state from a liquid to a gas.
Ø There is a difference between evaporation and the process of boiling. In evaporation the liquid molecules escape without the external heating source but in boiling a heat source is applied so the molecules get the energy to escape.
Effect of impurities on the melting and boiling points
Expansion in Liquids
Ø Liquids expand in the same way as solids but the expansion is ten to thirty times greater than of solids.
Ø Water expands in a different way. Water contracts as it cools down from 100°C to 4°C. However between 4°C and 0°C it expands at a very fast rate. Therefore the maximum density of water is at 4°C.
Transfer of Heat energy
Ø Heat energy can be transmitted form a hotter body to a cooler body till the thermal equilibrium is reached.
Ø Heat can be transmitted by Conduction, Convection and Radiation.
- Conduction is the flow of heat through a body from places of higher temperature to places of lower temperature.
Good Conductors of heat are used whenever heat is required to travel quickly.
Bad Conductors of heat are used where no heat is needed. Handles of kettles, saucepans etc.
- Convection is a much quicker means of heat transfer in gases and liquids then conduction. In convection the actual material moves, carrying heat with it.
- Radiation is defined as the mode of transfer of heat in the form of electromagnetic waves. In radiation no medium is required.
Ø Liquids expand in the same way as solids but the expansion is ten to thirty times greater than of solids.
Ø Water expands in a different way. Water contracts as it cools down from 100°C to 4°C. However between 4°C and 0°C it expands at a very fast rate. Therefore the maximum density of water is at 4°C.
Transfer of Heat energy
Ø Heat energy can be transmitted form a hotter body to a cooler body till the thermal equilibrium is reached.
Ø Heat can be transmitted by Conduction, Convection and Radiation.
- Conduction is the flow of heat through a body from places of higher temperature to places of lower temperature.
Good Conductors of heat are used whenever heat is required to travel quickly.
Bad Conductors of heat are used where no heat is needed. Handles of kettles, saucepans etc.
- Convection is a much quicker means of heat transfer in gases and liquids then conduction. In convection the actual material moves, carrying heat with it.
- Radiation is defined as the mode of transfer of heat in the form of electromagnetic waves. In radiation no medium is required.